2019 Volume No 38 – pages 35-50
Title: Characterisation and in vitro and in vivo evaluation of supercritical-CO2-foamed β-TCP/PLCL composites for bone applications |
Authors: S Pitkänen, K Paakinaho, H Pihlman, N Ahola, M Hannula, S Asikainen, M Manninen, M Morelius, P Keränen, J Hyttinen, M Kellomäki, O Laitinen-Vapaavuori, S Miettinen |
Address: Faculty of Medicine and Health Technology, FI-33014 Tampere University, Tampere, Finland |
E-mail: sanna.pitkanen at tuni.fi |
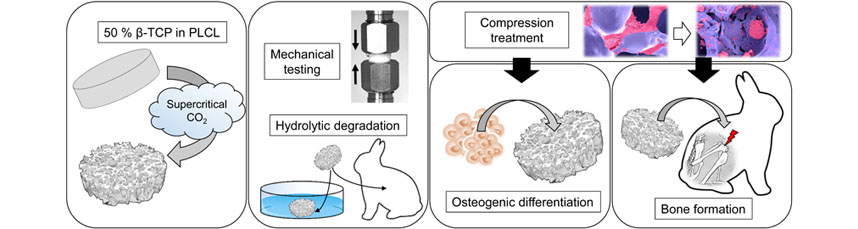
Abstract: Most synthetic bone grafts are either hard and brittle ceramics or paste-like materials that differ in applicability from the gold standard autologous bone graft, which restricts their widespread use. Therefore, the aim of the study was to develop an elastic, highly porous and biodegradable β-tricalciumphosphate/poly(L-lactide-co-ε-caprolactone) (β-TCP/PLCL) composite for bone applications using supercritical CO2 foaming. Ability to support osteogenic differentiation was tested in human adipose stem cell (hASC) culture for 21 d. Biocompatibility was evaluated for 24 weeks in a rabbit femur-defect model. Foamed composites had a high ceramic content (50 wt%) and porosity (65-67 %). After 50 % compression, in an aqueous environment at 37 °C, tested samples returned to 95 % of their original height. Hydrolytic degradation of β-TCP/PLCL composite, during the 24-week follow-up, was very similar to that of porous PLCL scaffold both in vitro and in vivo. Osteogenic differentiation of hASCs was demonstrated by alkaline phosphatase activity analysis, alizarin red staining, soluble collagen analysis, immunocytochemical staining and qRT-PCR. In vitro, hASCs formed a pronounced mineralised collagen matrix. A rabbit femur defect model confirmed biocompatibility of the composite. According to histological Masson-Goldner’s trichrome staining and micro-computed tomography, β-TCP/PLCL composite did not elicit infection, formation of fibrous capsule or cysts. Finally, native bone tissue at 4 weeks was already able to grow on and in the β-TCP/PLCL composite. The elastic and highly porous β-TCP/PLCL composite is a promising bone substitute because it is osteoconductive and easy-to-use and mould intraoperatively. |
Key Words: Bone substitute, composite, adipose stem cells, osteogenic differentiation, rabbit distal femur defect, β-tricalciumphosphate, poly(L-lactide-co-ε-caprolactone). |
Publication date: August 5th 2019 |
Article download: Pages
35-50 (PDF file) |