2021 Volume No 41 – pages 421-430
Title: Phagocytosis and macrophage polarization on bacterially contaminated dental implant materials and effects on tissue integration |
Authors: Y Yuan, Y Ren, M Dijk, GI Geertsema-Doornbusch, J Atema-Smit, HJ Busscher, HC van der Mei |
Address: Biomedical Engineering, University Medical Centre
Groningen, Antonius Deusinglaan 1, 9713 AV Groningen, the Netherlands |
E-mail: h.c.van.der.mei at umcg.nl |
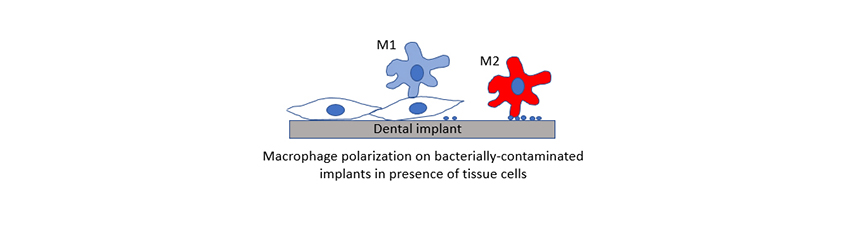
Abstract: Bacterial contamination is hard to avoid during dental implant surgery. Macrophages and their polarisation play a decisive role in bacterial colonisation and tissue integration on bacterially contaminated dental implants. The present study investigated the role of macrophages in stimulating tissue coverage overgrowth of contaminating oral bacteria on polished titanium (Ti-P) and acid-etched zirconium dioxide (ZrO2-MA) dental implant materials. Different co-culture models were employed to determine phagocytosis rates of Streptococcus mitis or Staphylococcus aureus contaminating a dental implant surface and the influence of contaminating bacteria and osteoblasts (U2OS) on macrophage polarisation. S. aureus was phagocytized in higher numbers than S. mitis in bi-cultures on smooth Ti-P surfaces. Contaminating S. mitis stimulated near full polarisation of macrophages from a non-Ym1-expressing- to a Ym1-expressing-phenotype on smooth Ti-P, but on ZrO2-MA both phenotypes occurred. In tri-cultures with U2OS-cells on smooth Ti-P, a larger percentage of macrophages remained in their non-Ym1-expressing, “fighting” M1-like phenotype to clear Ti-P surfaces from contaminating bacteria. On ZrO2-MA surfaces, more macrophages tended towards their “fix- and-repair” M2-like phenotype than on Ti-P surfaces. Surface coverage of smooth, bacterially contaminated Ti-P surfaces by U2OS-cells was more effectively stimulated by fighting, M1-like macrophages than on ZrO2-MA surfaces. Comprehensive guidelines are provided for the development of infection-resistant, dental implant materials, including bacteria, tissue and immune cells. These guidelines point to more promising results for clinical application of Ti-P as compared with ZrO2-MA. |
Key Words: Titanium, zirconium, M1 phenotype, M2 phenotype, co-cultures, U2OS cells, YM1, bacterial
adhesion. |
Publication date: March 30th 2021 |
Article download: Pages
421-430 (PDF file) |