2022 Volume No 43 – pages 130-152
Title: Regulators of collagen crosslinking in developing and adult tendons |
Authors: AJ Ellingson, NM Pancheri, NR Schiele |
Address: Chemical and Biological Engineering, University of Idaho, 875 Perimeter Dr. MS 0904, Moscow, ID, USA
|
E-mail: gaskon.ibarretxe at ehu.eus |
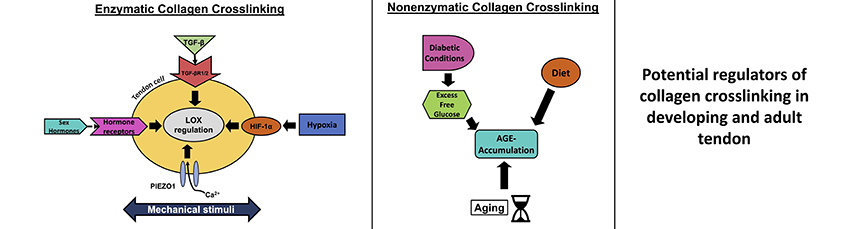
Abstract: Tendons are collagen-rich musculoskeletal tissues that possess the mechanical strength needed to transfer forces between muscles and bones. The mechanical development and function of tendons are impacted by collagen crosslinks. However, there is a limited understanding of how collagen crosslinking is regulated in tendon during development and aging. Therefore, the objective of the present review was to highlight potential regulators of enzymatic and non-enzymatic collagen crosslinking and how they impact tendon function. The main collagen crosslinking enzymes include lysyl oxidase (LOX) and the lysyl oxidase-like isoforms (LOXL), whereas non-enzymatic crosslinking is mainly mediated by the formation of advanced glycation end products (AGEs). Regulators of the LOX and LOXL enzymes may include mechanical stimuli, mechanotransducive cell signaling pathways, sex hormones, transforming growth factor (TGF)β family, hypoxia, and interactions with intracellular or extracellular proteins. AGE accumulation in tendon is due to diabetic conditions and aging, and can be mediated by diet and mechanical stimuli. The formation of these enzymatic and non-enzymatic collagen crosslinks plays a major role in tendon biomechanics and in the mechanisms of force transfer. A more complete understanding of how enzymatic and non-enzymatic collagen crosslinking is regulated in tendon will better inform tissue engineering and regenerative therapies aimed at restoring the mechanical function of damaged tendons. |
Keywords: Tendon, development, collagen, crosslinking, advanced glycation end products (AGEs), lysyl oxidase (LOX). |
Publication date: April 5th 2022 |
Article download: Pages
130-152 (PDF file) |