2024 Volume No 47 – pages 170-189
Title: A roadmap of osteogenic differentiation in human mesenchymal stem cells assessed by protein multiplex analysis |
Authors: M Eischen-Loges, Z Tahmasebi Birgani, Y Alaoui Selsouli, L Eijssen, H Rho, M Sthijns, M van Griensven, V LaPointe, P Habibović |
Address: Vanessa LaPointe, MERLN Institute for Technology-Inspired Regenerative Medicine, Maastricht University, 6200 MD, Maastricht, the Netherlands; Pamela Habibovic, MERLN Institute for Technology-Inspired Regenerative Medicine, Maastricht University, 6200 MD, Maastricht, the Netherlands |
E-mail: v.lapointe at maastrichtuniversity.nl; p.habibovic at maastrichtuniversity.nl |
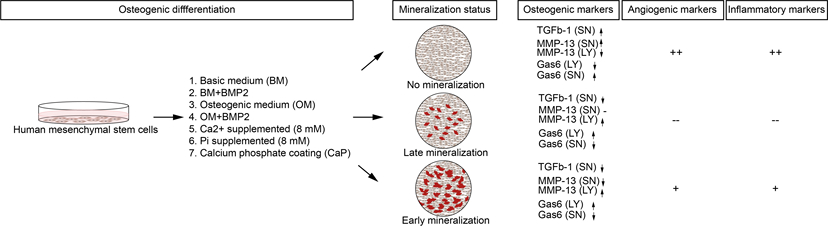
Abstract: Human mesenchymal stem cells (hMSCs) are widely used to model osteogenic differentiation in vitro, yet few studies compare the numerous available protocols. The poor translation between in vitro and in vivo results in bone regeneration highlights the need for improved methodologies to assess osteogenic phenotype. Omics technologies generate vast biological data but are complex for comparing several osteogenic differentiation strategies. Conversely, conventional methods like Alizarin red S stains or alkaline phosphatase assays do not provide enough data. Here, we propose a targeted protein multiplex assay to characterize and compare several models of osteogenic differentiation in hMSCs, containing markers for osteogenesis, angiogenesis, and inflammation—critical processes in bone regeneration. To induce osteogenic differentiation, hMSCs were stimulated with dexamethasone, with bone morphogenetic protein 2, supplemented with calcium or phosphate ions, or seeded on a calcium phosphate–based coating. Based on mineralization status determined by Alizarin red S stain, conditions were classified into non-mineralizing, late-mineralizing, and early-mineralizing groups. Protein patterns associated with these groups revealed distinct mechanisms of osteogenic differentiation for early- and late-mineralizing hMSCs. While non-mineralizing hMSCs did not undergo osteogenic differentiation, they exhibited increased angiogenic and inflammatory marker expression compared to the control. Late-mineralizing hMSCs had limited potential to produce angiogenesis-related factors, while early-mineralizing hMSCs showed an increased effect. Overall, we compared commonly used in vitro models of osteogenic differentiation in hMSCs to establish a comprehensive roadmap of osteogenic differentiation. |
Keywords: Protein muliplex, human mesenchymal stem cells, osteogenic differentiation, calcium phosphate, mineralization. |
Publication date: May 30th 2024 |
Copyright policy: © 2024 The Author(s). Published by Forum Multimedia Publishing, LLC. This article is distributed in accordance with Creative Commons Attribution Licence (http://creativecommons.org/licenses/by/4.0/). |
Article download: Pages 170-189 (PDF file) |